2
6
1
Use of this Guidance
THE PRIOR INFORMED CONSENT (PIC) PROCEDURE
INTRODUCTION AND SUMMARY
COORDINATION WITH KEY STAKEHOLDERSAT THE NATIONAL LEVEL
3
7
INFORMATION EXCHANGE
ANNEXES
5
EXPORT NOTIFICATION AND INFORMATIONTO ACCOMPANY EXPORT
RESPONSIBILITIES OF DNAS AND GOVERNMENTSIN IMPLEMENTING THE ROTTERDAM CONVENTION
4
CHEMICALS IN THE ROTTERDAM CONVENTION
1.2 Origins of the Rotterdam Convention
1.4.1 The Prior Informed Consent (PIC) Procedure
1.4.4 Key Players
1.3.4 Relationship to other “Chemicals Conventions”
1.3.1 Objective
1.3 What is the Rotterdam Convention?
1.4.3 Key Documents
1.2.3 Process for the Development of the Convention
1.2.2 Original Prior Informed Consent (PIC) Procedure
1.2.4 Negotiation Process and Interim Arrangements
1.3.3 What the Convention is and What the
Convention is not
1.1 Introduction and Purpose of this Guidance Document
1.4 How the Rotterdam Convention Operates – Key Elements
1.4.2 Information Exchange
1.2.1 Code of Conduct and London Guidelines
1.3.2 Scope - Chemicals covered
1.3.3 What the Convention is and What the
Convention is not
2.2.1 Which Chemicals are included
2.4 How Chemicals are removed from Annex III of the Rotterdam Convention
2.3.2 National Action - Proposals for Severely
Hazardous Pesticide Formulations
2.2.2 Which Chemicals are excluded
2.1 Introduction
2.3.3 Global Coordination - Role of the Secretariat,
Chemical Review Committee (CRC) and Conference of the Parties
2.3 How Chemicals are Added to Annex III of the Rotterdam Convention
2.2 Scope
2.3.1 National Action - Notifications of Final Regulatory
Action to Ban or Severely Restrict a Chemical
3.1 Introduction
3.3.3 Importing Parties
3.2 What is the Prior Informed Consent (PIC) Procedure?
3.3.4 Exporting Parties
3.3.1 PIC Circular
3.4 How the Prior Informed Consent (PIC) Procedure benefits Parties
3.3 How the PIC Procedure Works –Key Tools
3.3.2 Decision Guidance Document (DGD)
4.2 How the Export Notification Process Works
4.3 How Export Notification Benefits Parties
4.4 Information to accompany Exports
4.1 Introduction
4.5 How Information to Accompany Exports Benefits Parties
5.1 Introduction
5.2.1 Key provisions of information exchange
5.2.2 Key tools of information exchange
5.2 How Information Exchange Works
5.3 How Information Exchange Benefits Parties
6.3 Other obligations for Parties
6.4 Technical Assistance
6.1 Getting Started
6.2 Responsibilities of the Designated National Authority (DNA)
6 Checklist of actions
7.2 Elements relevant to the Customs officials
7.1 Government stakeholders
7.3 Non-Governmental Stakeholders
7 Coordination with key stakeholders at national level
1.3.3 What the Convention is and What the
Convention is not
________
____
1. Introduction and summary
1.1 Introduction and Purpose of this Guidance Document
The text of the Rotterdam Convention on the Prior Informed Consent (PIC) Procedure for Certain Hazardous Chemicals and Pesticides in International Trade was adopted at the Conference of Plenipotentiaries in Rotterdam on 10 September 1998. The Convention entered into force on 24 February 2004. For those unfamiliar with the details of the Convention, the obligations on its Parties and the operation of the Convention may seem complex and difficult to implement.
The primary purpose of this guide is to provide guidance to Designated National Authorities (DNAs) on the rights and obligations of Parties under the Convention.
To facilitate the work of the DNAs, the guide includes:
Background and contextual information to countries on the purpose and function of the Rotterdam Convention.A step–by–step, easy to follow explanation of the responsibilities of countries as Parties to the Convention. Each section of the guide may be used separately; as such, there is some redundancy between sections.A checklist which acts as a guide to actions that are required under the Convention.The appendices to the guide include the text of the Convention and forms and instructions developed in support of the key operational articles of the Convention (Annexes 3 and 4). The annexes are included in order for the guide to provide a comprehensive reference on the implementation of the Rotterdam Convention. A section on frequently asked questions (FAQs) has also been included, based on the experience of the Secretariat in working with Parties to implement the Convention (Annex 6). This guidance document, and in particular the "FAQs" section, is not intended to interpret the Convention but rather to provide simple guidance to governments. The Rotterdam Convention web site www.pic.int is an important source of current information on the operation and status of implementation of the Convention. Copies of all relevant forms and instructions, meeting reports etc., may be found therein
1.2 Origins of the Rotterdam Convention
1.2.1 Code of Conduct and London Guidelines
Increased production, trade and use of chemicals during the 1960s and 1970s coincided with a growing awareness of and concern about the risks that the use of hazardous chemicals could pose to human health and the environment. In addition, there were concerns that regulatory action in some Parties to ban or restrict the use of certain chemicals could result in these same chemicals being exported to other Parties where regulatory systems, infrastructure and resources were sometimes not adequate to assess and control the risks.
In response to these concerns FAO developed the voluntary International Code of Conduct on the Distribution and Use of Pesticides. The Code of Conduct was adopted in 1985, and subsequently amended in 1989, 2002 and 2013, to reflect changing trends in pest and pesticide management. In parallel with these initiatives, UNEP developed the London Guidelines for the Exchange of Information on Chemicals in International Trade to assist countries in managing the risks associated with industrial chemicals.
1.2.2 Original Prior Informed Consent (PIC) Procedure
In 1989, both the FAO Code of Conduct and the London Guidelines were amended to address issues related to the export of pesticides from a country that had itself domestically banned those pesticides. At that time, the Governing Bodies of FAO and UNEP agreed to work co-operatively and in 1992 implemented a joint programme on the Prior Informed Consent (PIC) Procedure. This became known as the original PIC procedure and remained in operation until the adoption of the text of the Rotterdam Convention in September 1998.
The original PIC procedure was supported by the FAO/UNEP Joint Group of Experts on Prior Informed Consent, established to provide technical assistance in the assessment of candidate chemicals. During this period, the Expert Group met eight times and agreed to include 17 pesticides, five severely hazardous pesticide formulations (SHPFs) and five industrial chemicals in the PIC procedure.
1.2.3 Process for the development of the Convention
In 1992, the United Nations Conference on Environment and Development (UNCED) recommended that the PIC procedure should be further developed into a legally-binding
instrument (Agenda 21, Chapter 19, paragraph 19.39d). Following this recommendation, the FAO Council agreed, at its 107th Session in November 1994, that “the Secretariat should proceed with the preparation of a draft legally-binding instrument on the operation of the PIC procedure as part of the joint FAO/UNEP programme on PIC and in cooperation with other international and non-governmental organizations concerned.” The Council also “agreed that the Organization should seek resources, jointly with UNEP, to finance the process for the preparation and conclusion of the legally binding instrument”. Subsequently the Governing Council of UNEP, at its 18th Session in May 1995, authorized the Executive Director to prepare for and convene, together with FAO, an intergovernmental negotiating committee. Its mandate was to prepare an international legally-binding instrument for the application of the PIC procedure for certain hazardous chemicals and pesticides in international trade (Decision GC 18/12).
1.2.4 Negotiation Process and Interim Arrangements
Commencing in March 1996, UNEP and FAO convened five meetings of the Intergovernmental Negotiating Committee (INC). Governments and inter-governmental and non-governmental organizations attended the negotiating sessions. The fifth and final negotiating session was held in Brussels, Belgium from 9 to 14 March 1998.
The text of the Convention agreed at the fifth session of the Intergovernmental Negotiating Committee was adopted on 10 September 1998 in Rotterdam, The Netherlands, as the "Rotterdam Convention on the Prior Informed Consent Procedure for Certain Hazardous Chemicals and Pesticides in International Trade". The Conference agreed that the 27 chemicals that were subject to the original PIC procedure should be included in Annex III of the Convention.
The Conference also adopted a resolution on interim arrangements so that the original PIC procedure might continue to operate, pending the entry into force of the Convention, and to prepare for its effective operation once it entered into force. The resolution made changes to the original PIC procedure to bring it in line with the provisions of the Convention. During the interim period, the PIC procedure was consequently known as the interim PIC procedure. During the interim period, any state or regional economic integration organization that nominated a designated national authority (DNA) was considered as participating in the interim PIC procedure. FAO and UNEP jointly provided the Secretariat.
The Rotterdam Convention entered into force on 24 February 2004, 90 days after the 50th instrument of ratification, acceptance or accession to the Convention was deposited in New York. The first meeting of the Conference of the Parties was convened in September 2004.
At its first meeting, the Conference of the Parties, in decision RC.1/13, adopted transitional arrangements which would allow non-Parties to continue participating in the interim PIC procedure for a period of two years, from the entry into force of the Convention to 24 February 2006. The same decision provided that at the end of the transition period, import responses, notifications and the list of DNAs from non-Parties should be retained, but not updated or circulated, by the Secretariat. That information is accordingly maintained on the Convention website.
Up-to-date information on states that are Party, chemicals included in Annex III and the status of implementation of the Convention can be found on the Convention website.
1.3 What is the Rotterdam Convention?
1.3.1 Objective
The overall objective of the Convention is to promote shared responsibility and cooperative efforts among Parties in the international trade of certain hazardous chemicals in order to protect human health and the environment from potential harm and contribute to the environmentally sound use of these chemicals.
1.3.2 Scope - Chemicals covered
The chemicals eligible for inclusion in the Rotterdam Convention are industrial chemicals and pesticides that have been banned or severely restricted nationally for health and environmental concerns. A group of pesticides called severely hazardous pesticide formulations (SHPFs) are also included. This latter group refers to pesticide formulations found to cause health or environmental problems under the conditions of use in a developing country or a country with an economy in transition.
Upon the adoption of the text of the Convention (September 1998), the 27 chemicals listed in Annex III of the Convention included 17 pesticides, five severely hazardous pesticide formulations and five industrial chemicals. Between September 1998 and entry into force of the Convention, 14 additional chemicals were identified for inclusion in the Convention and were subject to the interim PIC procedure. The first meeting of the Conference of the Parties formally listed the 14 chemicals in Annex III of the Convention. Parties have included additional chemicals to Annex III of the Convention and it is expected that chemicals will continue to be included. For a current list of the chemicals included in Annex III of the Rotterdam Convention consult the Convention web site www.pic.int or Appendix III of the most recent PIC Circular (see Section 3.3.1).
1.3.3 What the Convention is and What the Convention is not
What the Convention is
The Convention is a multilateral environmental agreement whose primary purpose is to protect human health and the environment. It does this by facilitating information exchange on chemicals that have been banned or severely restricted by one or more national governments as well as severely hazardous pesticide formulations (SHPFs) causing problems under the conditions of use in developing countries or countries with economies in transition.
The PIC procedure is an integral part of the Rotterdam Convention. For those chemicals listed in Annex III of the Convention, it provides countries with a means to assess the risks associated with their use and to decide whether or not they will allow future imports. It also provides a means for formally obtaining and disseminating the decisions of Parties regarding future imports of these chemicals. Exporting Parties are obliged to ensure that exports do not occur contrary to these decisions.
What the Convention is not
The listing of a chemical in Annex III of the Rotterdam Convention is not a recommendation to ban or severely restrict its use. The decision to include a chemical in Annex III is triggered by the final regulatory actions to ban or severely restrict a chemical by at least two Parties from two different PIC regions (see Section 1.4.4). Pesticide formulations causing problems under the conditions of use in a Party that is a developing country or country with an economy in transition may also be included. Parties to the Convention are expected to make their own informed decisions regarding the future import and use of chemicals listed in Annex III and subject to the PIC procedure. It is not intended that these chemicals be automatically subject to national regulatory actions to ban or severely restrict their use.
1.3.4 Relationship to other Conventions relating to chemicals
Other international conventions for the management of chemicals exist in tandem with the Rotterdam Convention. These include the Stockholm Convention on Persistent Organic Pollutants and the Basel Convention on the Control of Transboundary Movement of Hazardous Wastes and Their Disposal. These conventions are related in their aim to manage chemicals, but are distinct in scope, objectives and function.
Stockholm Convention on Persistent Organic Pollutants (POPs)
The Stockholm Convention on Persistent Organic Pollutants (POPs) seeks the elimination or restriction of production and use of all intentionally produced POPs (i.e. industrial chemicals and pesticides). It also seeks the continuing minimization and, where feasible, ultimate elimination of the releases of unintentionally produced POPs such as dioxins and furans.
Several pesticides and industrial chemicals included in the Stockholm Convention are also subject to the Rotterdam Convention, such as: polychlorinated biphenyls (PCBs), aldrin, dieldrin, DDT, chlordane, hexachlorobenzene, lindane, toxaphene and heptachlor. For a current list of the chemicals included in the Stockholm Convention consult the Convention web site www.pops.int.
It is important to recognize that chemicals are included in the Stockholm Convention based on a defined set of characteristics including their toxicity, potential for bio-accumulation, environmental persistence and transboundary movement to locations remote from their release. However, it should be noted that chemicals are not subject to the Stockholm Convention because of any national regulatory action to ban or severely restrict their use.
Chemicals that are subject to final regulatory actions to ban or severely restrict their use, demonstrating one or more of the characteristics stated above, would be candidates for inclusion in the Rotterdam Convention. Therefore, some chemicals will be subject to both Conventions. Ultimately it is expected that most, if not all, of the pesticides and industrial chemicals identified for inclusion in the Stockholm Convention will have already been subject to the Rotterdam Convention. Further information on the Stockholm Convention may be found at www.pops.int.
Basel Convention on the Control of Transboundary Movements of Hazardous Wastes and Their Disposal
The Basel Convention sets out a global mechanism for the control of the transboundary movement of hazardous and other wastes. It has similarities to the Rotterdam Convention in that it promotes information exchange and has provisions to control trade. The Rotterdam Convention specifically excludes wastes. Therefore, a chemical that has become a waste will fall within the scope of the Basel Convention rather than the Rotterdam Convention. Further information on the Basel Convention may be found at www.basel.int.
1.4 How the Rotterdam Convention Operates – Key Elements
1.4.1 The Prior Informed Consent (PIC) Procedure
The prior informed consent (PIC) procedure applies to the chemicals listed in Annex III of the Convention. For each of these chemicals a document, known as a Decision Guidance Document (DGD), is prepared and sent to all Parties with a request that they take a decision as to whether or not they will allow future import of this chemical. These decisions, known as import responses, are sent to the Secretariat. The Secretariat compiles these decisions and circulates them to all Parties every six months via the PIC Circular (see Section 3.3.1).
All Parties are required to ensure that exports of chemicals subject to the PIC procedure do not occur contrary to the decision of an importing Party. This means that an export must not be made to a Party that has indicated that it does not wish to receive imports of these chemicals. Similarly, if the importing Party has indicated that it will allow import subject to certain condition(s), then the exporting Party must ensure that exports to that importing Party are subject to these condition(s).
The key is that shipments of these chemicals from exporting Parties do not take place without the prior informed consent of the importing Party.
1.4.2 Information Exchange
The Convention contains several provisions pertaining to information exchange (see Section 5 Information Exchange). These include Parties receiving summaries of all notifications of final regulatory actions to ban or severely restrict chemicals as well as proposals for severely hazardous pesticide formulations (SHPFs). The Convention requires Parties that have banned or severely restricted a chemical in their own territory to notify individual importing Parties of the first proposed export in each calendar year (see section 4 Export Notification). Exports of such chemicals, as well as those subject to the PIC procedure (listed in Annex III of the Convention), are to be appropriately labelled and accompanied by basic health and safety information in the form of a safety data sheet. In addition, Parties have access to information about the DNA(s) in every other Party. The Convention also contains general obligations for the provision of information on chemicals to be made publicly available, as well as to be provided when requested by other Parties.
1.4.3 Key Documents
The Text of the Convention sets out the provisions and obligations that apply to all Parties. A copy of the full text of the Convention is included in Annex 3 to this document. Copies of the Text of the Convention in six languages (Arabic, Chinese, English, French, Russian and Spanish) can be downloaded from the Rotterdam Convention web site www.pic.int.
Forms and Instructions
The Secretariat has developed a set of forms, and accompanying instructions, to facilitate the implementation of the key operational articles of the Convention. These include:
Notification of Final Regulatory Actions form and instructions (Article 5);Import Response form and instructions (Article 10);Severely Hazardous Pesticide Formulations (SHPF) Incident Report forms (one for human poisoning incidents and one for environmental incidents) and instructions (Article 6);Export notification form (Article 12).The forms with instructions to complete them have been included in Annex 5 to this guide. An electronic version of the forms can be accessed from the Rotterdam Convention web site www.pic.int.
PIC Circular
The PIC Circular is one of the key documents in the operation of the Rotterdam Convention. It provides information to Parties on the status of implementation of the Convention and, in particular, responds to the requirements of Articles 4, 5, 6, 7, 10, 11 and 14 of the Convention regarding communications between the Secretariat and Parties.
The PIC Circular is distributed by the Secretariat to all Parties every six months, in June and December. It contains:
summaries of notifications of final regulatory actions for banned or severely restricted chemicals (Appendix I);summaries of proposals for severely hazardous pesticide formulations - SHPFs (Appendix II)a list of the chemicals included in Annex III and subject to the PIC procedure (Appendix III);a compilation of import decisions for all of the chemicals subject to the PIC procedure and a list of Parties that have failed to provide import decisions for chemicals subject to the PIC procedure (Appendix IV);a tabular summary of notifications of final regulatory action received for chemicals not included in Annex III but found to meet the requirements of Annex I of the Convention (Appendix V);information exchange on chemicals recommended by the Chemical Review Committee for listing in Annex III but for which the Conference of the Parties has yet to take a final decision (Appendix VI);other relevant information such as requests from a Party for information about the transit of chemicals included in Annex III of the Convention through its territory, information submitted to the Secretariat for circulation to Parties on domestic regulatory actions; and the status of ratification.A detailed outline of the contents of the PIC Circular is provided in Annex 2 to this guide.
Decision Guidance Documents (DGDs)
For each chemical listed in Annex III of the Convention and subject to the PIC procedure, a decision guidance document (DGD) is prepared. It contains basic information on the chemical, such as its hazard classification, additional sources of information on the chemical and information on possible alternatives.
For banned or severely restricted chemicals, the DGD contains details of the regulatory actions of the two or more Parties that led to the chemical being included in Annex III and a summary of the risk evaluation and supporting documentation considered in support of these regulatory actions.For SHPFs, the DGD includes a description of the common and recognized patterns of use of the formulation in the proposing country as well as the incidents related to the formulation that were considered in deciding to include it in Annex III.A decision guidance document is not intended as the only source of information on a chemical, nor is it updated or revised following its adoption. Additional information submitted by Parties for chemicals in Annex III is available on the Rotterdam Convention web site www.pic.int.
The PIC Circular, and all information it contains, is also available on the Convention website www.pic.int. A detailed outline of the contents of a decision guidance document is provided in Annex 1 to this guide.
1.4.4 Key Players
Parties and their Designated National Authorities (DNAs)
For the Rotterdam Convention, “Parties” are the countries or regional economic integration organizations that have ratified, accepted, approved or acceded to the Convention.
As set out in Article 4, Parties are required to nominate one or more DNA(s), who are the primary contact point(s) for matters related to the operation of the Convention and who are authorized to perform the administrative functions necessary for the operation of the Convention. For a current list of the Parties to the Rotterdam Convention and the contact details for their respective DNAs, consult the Rotterdam Convention web site www.pic.int.
Secretariat
The Secretariat for the Rotterdam Convention is provided jointly by FAO and UNEP. The functions of the Secretariat are outlined in Article 19 of the Convention. Briefly, the Secretariat is responsible for making the administrative arrangements for meetings of the Conference of the Parties and its subsidiary bodies, disseminating import responses received from Parties for chemicals in Annex III of the Convention, verifying that information in support of notifications of final regulatory actions and proposals for severely hazardous pesticide formulations has been provided, facilitating assistance to developing country Parties in the implementation of the Convention and information exchange between Parties as well as ensuring coordination with the secretariats of other relevant international bodies.
Conference of the Parties (COP)
The Conference of the Parties (COP) oversees the operation of the Convention and is comprised of Parties to the Convention. The reports of the COP, including its decisions, can be found on the Convention web site www.pic.int.
Chemical Review Committee (CRC)
Article 18 of the Convention provides for the establishment of a subsidiary body called the Chemical Review Committee (CRC). This Committee reviews the relevant supporting information for candidate chemicals, including severely hazardous pesticide formulations, in line with the information requirements and criteria set out in the Convention. The CRC recommends to the Conference of the Parties whether or not a chemical should be listed in Annex III of the Convention and subject to the PIC procedure. The membership of the CRC is composed of 31 government-designated experts in chemical management from the five UN Regions. The members of the CRC are appointed by the COP. For further information on the operation of the CRC and the process by which chemicals are added to Annex III, see Section 2.3 of this guide. The reports of the CRC, including its recommendations on candidate chemicals, can be found on the Rotterdam Convention web site www.pic.int.
Prior Informed Consent (PIC) Regions
Article 5 of the Convention requires that, when the Secretariat has received at least one notification of final regulatory action from each of two PIC Regions regarding a particular chemical, it shall forward them to the Chemical Review Committee. The Convention recognizes seven PIC regions (Africa, Asia, Europe, Latin America and the Caribbean, Near East, North America and Southwest Pacific) and countries have been assigned to those regions. For information on the composition of the Prior Informed Consent Regions, consult the Rotterdam Convention web site www.pic.int.
2. Chemicals in the Rotterdam Convention
2.1 Introduction
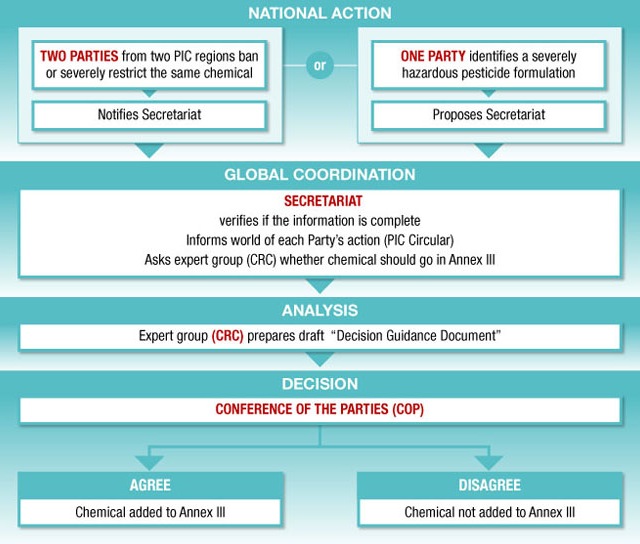
This section highlights the broad range of chemicals that are covered by the two key provisions of the Convention: (i) information exchange, (ii) the PIC procedure. It provides a detailed explanation of the process for the addition of chemicals to the PIC procedure and Annex III of the Convention. A schematic diagram of the process and a simple summary of the individual steps are provided in Figure 1. This section also describes the roles and responsibilities of DNA(s) at the national level, and the Secretariat, Chemical Review Committee (CRC) and Conference of the Parties (COP) at the global level.
2.2 Scope
2.2.1 Which Chemicals are included
The information exchange provisions of the Convention apply broadly to any chemical that has been banned or severely restricted to protect human health or the environment by a Party, or to severely hazardous pesticide formulations causing problems under the conditions of use in a developing country Party or Party with an economy in transition. The provisions of the prior informed consent (PIC) procedure apply only to those chemicals listed in Annex III of the Convention.
Article 2 defines the following terms for the purposes of the Convention:
”Chemical” means a substance whether by itself or in a mixture or preparation and whether manufactured or obtained from nature but does not include a living organism. It consists of the following categories: pesticides (including severely hazardous pesticide formulations) and industrial chemicals.
“Banned chemical” means a chemical all uses of which within one or more categories, have been prohibited by final regulatory action in order to protect human health or the environment. It includes a chemical that has been refused approval for first-time use or has been withdrawn by industry, either from the domestic market or from further consideration in the domestic approval process, and where there is clear evidence that such action has been taken in order to protect human health or the environment.
“Severely restricted chemical” means a chemical virtually all use of which, within one or more categories, has been prohibited by final regulatory action in order to protect human health or the environment, but for which certain specific uses remain allowed. It includes a chemical that has, for virtually all use, been refused approval or been withdrawn by industry either from the domestic market or from further consideration in the domestic approval process, and where there is clear evidence that such action has been taken in order to protect human health or the environment.
“Severely hazardous pesticide formulations” (SHPF) means a chemical formulated for pesticidal use that produces severe health or environmental effects observable within a short period of time after single or multiple exposure, under conditions of use.
2.2.2 Which Chemicals are excluded
Further clarification of the scope of the Convention is provided in Article 3 which specifies the sort of chemicals excluded from the Convention. These include: narcotic drugs and psychotropic substances; radioactive materials; wastes; chemicals used in chemical weapons; human or veterinary drugs; food additives and food itself. For the most part these items are the subject of other international agreements. For example, the Codex Alimentarius covers food additives and residues of pesticides in food, while the Basel Convention covers wastes.
The Convention does not cover small quantities of pesticides or industrial chemicals that are not likely to affect human health or the environment, provided that they are intended to be used for research or analysis or by a person for their own personal use. These quantities are not defined in the Convention. Some Parties in implementing the Convention have set a level of 10 kilograms whereas others have set lower amounts. Whatever the amounts countries elect to apply, it is important to recognize that these should be small amounts compared to commercially traded quantities.
2.3 How Chemicals are Added to Annex III of the Rotterdam Convention
There are two principal means through which new chemicals are identified for inclusion in Annex III of the Convention. These are:
(1) notification by Parties of final regulatory actions to ban or severely restrict a chemical for health or environmental concerns;The obligations of Parties and the process for submissions and review of notifications of final regulatory actions are contained in Article 5 of the Convention. Annex I of the Convention details the information requirements and, Annex II the criteria to be considered by the Chemical Review Committee in reviewing candidate chemicals for inclusion in Annex III of the Convention.(2) a proposal from a Party which is a developing country or a country with an economy in transition that is experiencing human health or environmental problems with a severely hazardous pesticide formulation (SHPF) under the conditions of use in its territory;The process for the submission of proposals for SHPFs are contained in Article 6 of the Convention. Parts 1 and 3 of Annex IV of the Convention detail respectively the relevant supporting information required and the criteria considered by the Chemical Review Committee in reviewing candidate formulations for inclusion in Annex III of the Convention.2.3.1 National Action - Notifications of Final Regulatory Actions to ban or severely restrict a chemical
What is a banned chemical or a severely restricted chemical?The terms “banned chemical” and “severely restricted chemical” are defined in Article 2 of the Convention. Simply put:
a ban is where all uses of the chemical as a pesticide and/or an industrial chemical are prohibited;a severe restriction is where virtually all uses of a chemical as a pesticide and/or an industrial chemical have been prohibited but for which certain uses are still permitted.These definitions include situations where a chemical is prohibited for further use, where the chemical has been refused registration (or approval) for first time use, where industry withdraws its application for approval prior to the government making a final decision on the application or where it is withdrawn by industry from the domestic market. To qualify for notification under the Convention, these bans or severe restrictions must have been made for human health or environmental reasons.
It is relatively easy to determine when a final regulatory action is a ban, but it is sometimes more difficult to determine when a final regulatory action is a severe restriction. Where some uses have been prohibited, judgment is needed as to whether this constitutes a prohibition of virtually all uses. If all but one or two uses of an extensive range of uses have been prohibited and those remaining are relatively small uses, then this clearly constitutes a severe restriction. However, where all but one or two uses of an extensive range of uses have been prohibited and those remaining are major uses, then this may not constitute a severe restriction, particularly if the prohibited uses were all moderate to minor uses.
It is not uncommon for industry to withdraw applications for approval for uses or to withdraw already approved uses when it becomes apparent that there is a problem with these uses. In such circumstances, it may be difficult to determine whether this action has been taken because of commercial reasons or whether it is because industry is aware of the health or environmental concerns.
Obligations under Article 5 of the ConventionUnder Article 5 of the Convention, Parties have the following obligations with respect to notifying the Secretariat of their final regulatory actions to ban or severely restrict a chemical for health or environmental reasons:
On the entry into force of the Convention for a Party, the Party is obliged to notify the Secretariat of all its final regulatory actions (bans and severe restrictions) in effect at that time. Where Parties have made notifications under the original PIC procedure they do not need to resubmit these notifications.Parties have an ongoing obligation to notify the Secretariat of any subsequent final regulatory actions (bans or severe restrictions) taken for health or environmental reasons. These notifications should be submitted to the Secretariat by the DNA as soon as possible after the action has been taken, but no later than 90 days after the regulatory action has taken effect.Where a Party modifies a regulatory action notified previously to the Secretariat, the DNA should resubmit a notification reflecting the changed regulatory action.Some Parties may ban or severely restrict a chemical included in Annex III of the Convention. Parties are still obliged to submit notifications of these regulatory actions. The basis for a country’s regulatory action on a chemical may not be the same as that which led to the inclusion of the chemical in Annex III of the Convention and this could also lead to a broader scope of the chemical to be subject to the Convention . For example:the original reason for inclusion may have been related to environmental concerns whereas the more recent regulatory action to ban may be based on human health concerns;certain severely hazardous formulations of a chemical may be included in the Convention, however subsequent regulatory actions by Parties may, for example, ban all formulations of the chemical as a pesticide. This could lead to a broader scope of the chemical to be subject to the Convention;a chemical may be included in the Convention for one category, such as pesticide, however subsequent regulatory actions by Parties may, for example, apply to the another use category of the chemical, such as industrial chemical.
Process for submission of notification of a final regulatory actionAs set out in Article 5 of the Convention, notifications of final regulatory actions must contain the information specified in Annex I of the Convention, where it is available. The notification should describe the scope of the regulatory action, the categories and/or uses to which the action applies, the chemical concerned and details of the regulatory decision. The notification should also include the reason a decision was taken and whether it was based upon a risk or hazard evaluation. If the final regulatory action was based on a risk evaluation involving prevailing conditions in the notifying country, this should be indicated, and a summary of relevant information provided. Further supporting documentation can be submitted separately if available.
It should be noted that risk evaluation in the context of the Rotterdam Convention is neither hazard assessment nor risk assessment, but something in between. Risk evaluation considers information on hazard and exposure. In notifications of final regulatory action, information on hazards is often taken from internationally accepted toxicological or ecotoxicological sources, such as World Health Organization (WHO) publications or the Pesticide Manual. However, information on exposure is to be related to the prevailing conditions of use in the notifying country. It may take the form of actual or measured exposure, or reflect the results of modelling of expected or anticipated exposure.
To facilitate the preparation and submission of these notifications of final regulatory action a detailed form has been developed that aligns to the information requirements of Annex I of the Convention. The form with instructions on how to complete it are included in Annex 7 of this guide or can be downloaded from the Rotterdam Convention web site www.pic.int.
2.3.2 National Action - Proposals for Severely Hazardous Pesticide Formulations
What is a Severely Hazardous Pesticide Formulation (SHPF)?The term “severely hazardous pesticide formulation” is defined in Article 2 of the Convention. Simply put, they are formulations that cause problems (severe health or environmental effects observable within a short period of time after single or multiple exposures) under the conditions of use in developing country Parties or Parties with economies in transition.
The provisions for severely hazardous pesticide formulations were included in the Convention in recognition that in some developing countries and countries with economies in transition the conditions are such that certain pesticide formulations cannot be used safely. These same formulations may be used safely in developed countries and as a result would not be identified as candidates for inclusion under Article 5 of the Convention.
Obligations under Article 6 of the ConventionUnder Article 6, any Party that is a developing country or country with an economy in transition that is experiencing problems caused by a SHPF, either due to human health or environmental problems in its territory, may propose to the Secretariat the inclusion of the formulation in the Convention. The proposals must contain the information specified in Part 1 of Annex IV of the Convention and be submitted by the DNA of that country to the Secretariat. In preparing such proposals, the DNA may draw upon technical expertise from any relevant source.
Process for the submission of a Proposal for a SHPFTo facilitate the development and submission of proposals in support of severely hazardous pesticide formulations two incident report forms have been developed, one for environmental incidents and a second for human health incidents.
These forms consist of two parts, Part A and Part B. Part A (Transmittal Form) is to be used by the DNA to transmit an incident report form to the Secretariat. Part B (Pesticide Incident Report Form) has been developed to meet the information requirements of Part 1 of Annex IV of the Convention. It is intended to provide a clear description of the incidents related to the use of the pesticide formulation, any adverse effects and the way in which the formulation was used. The use of these forms is purely voluntary; other forms/formats used in a country for collecting pesticide incident reports may be used to replace Part B provided that those submissions meet the information requirements of Part 1 of Annex IV of the Convention.
Copies of the incident report forms and instructions on how to complete them are included in Annex 5 of this guide or can be downloaded from the Rotterdam Convention web site www.pic.int.
2.3.3 Global Coordination – Role of the Secretariat, Chemical Review Committee (CRC) and Conference of the Parties (COP)Role of the Secretariat: Verification of Notifications of Final Regulatory ActionWhen the Secretariat has received a notification for final regulatory action to ban or severely restrict a chemical, it must verify that the notification meets the information requirements of Annex I of the Convention. The Secretariat completes this review with the assistance of a detailed checklist. If the notification meets the information requirements, a draft summary is prepared. The notifying country is informed that their notification met the requirements and is invited to review the draft summary. The summaries of the verified notifications are published in Appendix I of the PIC Circular within six months of their being received.
If a notification does not meet the information requirements of Annex I of the Convention, the notifying country is informed and is provided with a completed checklist indicating the missing information. The Party is invited to submit the missing information so that the notification meets the requirements and, accordingly, a summary is prepared for publication in the PIC Circular.
When the Secretariat has received a notification for a chemical that is not listed in Annex III and has been verified as meeting the information requirements of Annex I, it requests the notifying Party to submit the supporting documentation referenced in the notification. Once the Secretariat has received two notifications for the same chemical verified as complete from at least two PIC Regions, the notifications and the supporting documentation are forwarded to the CRC for consideration.
Figure 1. Process for adding chemicals to Annex III of the ConventionRole of the Secretariat: Verification of Proposals for SHPFsWhen the Secretariat receives a proposal for a SHPF, it verifies that it includes the information specified in Part 1 of Annex IV of the Convention. If the submitted proposal meets the information requirements a draft summary is prepared. The proposing country is informed that their proposal was complete and invited to review the draft summary. The summaries of the verified proposals are published in Appendix II of the PIC Circular within six months of their being received.
At the same time, the Secretariat initiates collection of relevant information relating to the formulation as set out in Part 2 of Annex IV of the Convention. This includes information from other Parties, international organizations, non-governmental organizations or other relevant sources on handling restrictions or incidents related to the formulation and other formulations of the pesticide in question in other States, and risk or hazard evaluations. The proposal and the additional information collected by the Secretariat are forwarded to the CRC for consideration.
It is important to note that unlike notifications of final regulatory actions a proposal from one Party is all that is required to initiate review by the CRC.
Role of the Chemical Review Committee (CRC)After a proposal or notification and relevant supporting data are submitted to the CRC for their consideration, the process for the addition of a chemical to the Convention is the same. The primary difference remaining in the operation of the CRC is the criteria used to assess the different submissions. For a banned or severely restricted chemical the relevant criteria are contained in Annex II, whereas the criteria for severely hazardous pesticide formulations are contained in Part 3, Annex IV of the Convention.
Banned or severely restricted chemicalsIn reviewing notifications for banned or severely restricted chemicals, the CRC establishes that:
the action was taken to protect human health or the environment;the action was taken on the basis of a risk evaluation performed by the Party based on prevailing conditions within its territory;the final regulatory action has a significantly broad basis for listing in the Convention; andthere is continuing international trade in the chemical.Severely Hazardous Pesticide Formulations (SHPF)In reviewing the proposal for a SHPF, the CRC establishes whether:
the reported incidents were a result of the use of the pesticide formulation;the proposal has relevance to other Parties with similar climatic conditions and patterns of use of the formulation;severe handling restrictions for the chemical in other countries and the amount of chemical used in relation to the reported incident suggest that the chemical is highly hazardous.For both banned or severely restricted chemicals and severely hazardous pesticide formulations, the Convention states that intentional misuse (for example, deliberate ingestion as a means of suicide) in itself does not constitute adequate reason to list a chemical in Annex III.
If the CRC considers that the information in support of a banned or severely restricted chemical or a severely hazardous pesticide formulation meets the relevant information requirements and criteria set out in the Convention, it will recommend the inclusion of the chemical in Annex III of the Convention to the COP and initiate preparation of a draft decision guidance document (DGD) (see Section 3 for the process on developing a DGD).
The Chemical Review Committee has developed a number of papers with policy guidance and working procedures covering a broad range of issues related to the work of the Committee. Those documents are intended to facilitate the work of the Committee and help ensure consistency and transparency in its operation. They are revised, as necessary, in the light of experience. The policy guidance and working procedures are currently in use by the CRC and can be found on the Convention web site www.pic.int.
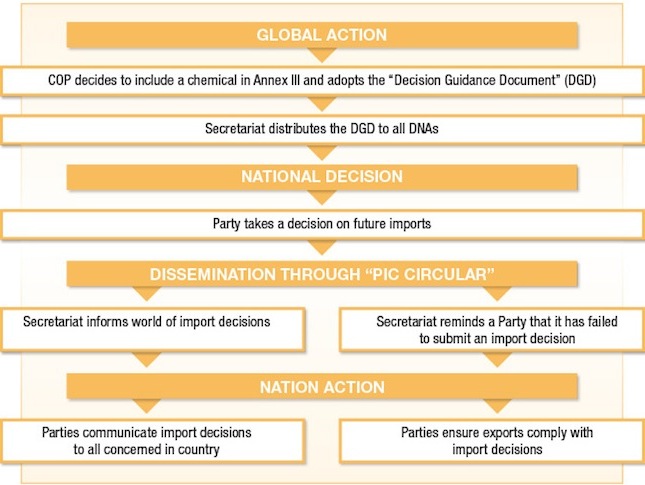
Role of the Conference of the Parties (COP)In line with Article 7 and Article 22 of the Convention, the Conference of the Parties (COP) will decide whether or not to include a chemical in Annex III of the Convention and, if so, to approve the draft decision guidance document. Once a decision to include a chemical in Annex III of the Convention is made, the Secretariat will circulate the decision and the approved decision guidance document to all Parties with a request that they provide a decision on future imports of the chemical.
2.4 How Chemicals are removed from Annex III of the Rotterdam Convention
Article 9 of the Convention addresses the removal of chemicals from Annex III of the Convention. A chemical may be considered for removal if a Party submits information which was not available when a decision was made to include a chemical in the Convention and that information indicates that the basis for inclusion of the chemical is no longer in accordance with the relevant.The Secretariat will forward the information to the CRC, which will review the information according to the relevant criteria. For each chemical that the CRC decides to recommend to the COP for removal, it shall prepare a revised draft decision guidance document. The COP then will decide upon the recommendation. If the COP decides to remove the chemical from Annex III of the Convention and approve the revised decision guidance document explaining why this chemical has been removed, the Secretariat will circulate this information to all Parties.
2.2.2 Which Chemicals are excluded
2.3.1 National Action - Notifications of Final Regulatory
Action to Ban or Severely Restrict a Chemical
2.4 How Chemicals are removed from Annex III of the Rotterdam Convention
2.3.2 National Action - Proposals for Severely
Hazardous Pesticide Formulations
2.3.3 Global Coordination - Role of the Secretariat,
Chemical Review Committee (CRC) and Conference of the Parties
3. The Prior Informed Consent (PIC) procedure
3.1 Introduction
The Prior Informed Consent or PIC procedure is the central core around which the Rotterdam Convention has been developed. This section of the document includes a brief overview of the PIC procedure as well as detailed information on how the process works, the relevant documents, the importance of import decisions and the roles and responsibilities of Parties with respect to the import and export of chemicals included in Annex III of the Convention.
This section summarizes the key provisions of the Convention concerning the PIC procedure and the key tools and mechanisms that have been developed to implement those provisions.
3.2 What is the Prior Informed Consent (PIC) Procedure?
For each chemical included in Annex III of the Convention, the PIC procedure applies. For each of these chemicals, a Decision Guidance Document is prepared and sent to all Parties with a request that they take a decision as to whether they will allow future import of the chemical. The resulting import decisions are published by the Secretariat and made available to all other Parties through the PIC Circular (see Sections 1.4.3 and 3.3.1 and Annex 2 to this guide).
The PIC procedure provides all Parties with an opportunity to make an informed decision as to whether they wish to receive future imports of the chemicals listed in Annex III of the Convention. All Parties are required to ensure that their exports do not take place contrary to an importing Party's import decision. The key action is that shipments of these chemicals from exporting Parties do not take place without the prior informed consent of the importing Party.
3.3 How the PIC Procedure Works – Key Tools
3.3.1 PIC Circular
The PIC Circular is compiled by the Secretariat and sent to DNAs every six months, in June and December. It provides information to Parties on the status of implementation of the Convention and, in particular, responds to the requirements of Articles 4, 5, 6, 7, 10, 11 and 14 of the Convention regarding communications between the Secretariat and Parties. It contains, among other things, summaries of notifications of final regulatory actions to ban or severely restrict chemicals and proposals for the inclusion of severely hazardous pesticide formulations.
Appendices III and IV of the PIC Circular are particularly relevant to the PIC procedure. Appendix III contains a current list of the chemicals subject to the PIC procedure. Appendix IV contains all the import decisions for chemicals subject to the PIC procedure that have been submitted by Parties as well as a list of those Parties that have failed to provide import decisions.
The compilation of import decisions in Appendix IV of the PIC Circular represents the official reference for Parties that are looking to export chemicals subject to the Convention.
A summary of the information contained in the PIC Circular is provided in Annex 2 to this guide.
3.3.2 Decision Guidance Document
A Decision Guidance Document has been developed for each of the chemicals included in Annex III of the Convention. The decision guidance document sets out the scope of the final regulatory actions for the chemicals subject to the PIC procedure, it provides basic information about the chemical's characteristics and summarizes the reasons why the chemical was either banned or severely restricted or, in the case of a SHPF, the problems found under the conditions of use.
A decision guidance document is not intended to be a scientific treatise on a given chemical but rather to assist governments in making an informed decision regarding future importation of the chemical. Governments may wish to seek further information, if they consider it necessary, before making an import decision.
A summary of information contained in a decision guidance document is provided in Annex 1 of this guide.
Chemicals included in Annex III of the Convention are those that Parties have determined, under the conditions of use in their territories, to pose unacceptable risks for all or some uses of these chemicals. These decisions and the underlying circumstances may have been specific to the proposing countries’ conditions of use and may or may not be related to the way in which the chemical is used in other countries or regions.
This means that Parties should consider the relevance of their national conditions with regard to the use of the chemical and take into account the information in the decision guidance document, on the basis of the reported regulatory actions to ban or severely restrict the chemical or the conditions under which a severely hazardous pesticide formulation was used, when making their decision as to the future import of the chemical.
When reviewing a decision guidance document in order to make a national decision for future import of an Annex III chemical, some of the factors to consider include:
whether the chemical is used in your country and, if so,consider how it is used;estimate the likely exposure of the chemical to workers, the public and non-target plants and animals; andassess whether this is similar to the conditions reported by the notifying or proposing Parties as the basis for their regulatory action.whether the climatic, environmental and other conditions are similar between your country and the notifying Party(ies);whether the level of chemicals management in your country is comparable to that of the notifying Party(ies); andif the notification was from a developed Party, whether that Party already had safety directions and precautions that are either not available in your country or impractical to use in your circumstances.Decision guidance documents often also contain information on alternative chemicals, if alternatives are available and if indicated in the notifications received. An important consideration when making a national decision may be whether there are safer alternative chemicals or techniques available that can replace the chemical in question. Particularly for pesticides, Parties should be aware that an alternative chemical identified by a notifying Party may not be an effective alternative under the conditions of use in your country. Parties may require data that demonstrates that the proposed alternative is effective and has no unacceptable effects on human health and the environment under the conditions of use in their country.
Parties should not limit their consideration of whether or not to import a chemical to the availability of alternatives for that chemical. For instance, the practice of integrated pest management (IPM) may include any combination of physical treatments, changes in management and cultural practices while minimizing the use of chemical treatments.
Decision guidance documents for all the chemicals included in Annex III of the Convention are available on the Convention website www.pic.int. It is important to note that decision guidance documents are not updated once they are adopted and reflect the information available at that time.
3.3.3 Importing Parties: Obligations and procedures for Annex III chemicals
Obligations under Article 10 of the Convention
Under Article 10 of the Convention, Parties have the following obligations with respect to imports of chemicals listed in Annex III of the Convention and subject to the PIC procedure:
No later than the date of entry into force of the Convention for a Party it is obliged to send to the Secretariat its decisions on future import for each of the chemicals listed in Annex III of the Convention. Where Parties have provided such import responses under the original voluntary PIC procedure they do not need to resubmit them.Parties have an ongoing obligation to submit to the Secretariat, as soon as possible and in any event no later than nine months after the date of dispatch of a decision guidance document, their import decision (whether a final or interim response) concerning the future import of the chemical.Where a Party changes an import decision submitted previously to the Secretariat, the DNA should submit a revised import response to the Secretariat as soon as possible.Types of responses
An import response shall consist of either a final decision based on national legislative or administrative measures or an interim response.
A final decision may include:
consent to future importation of the chemical; orno consent to future importation of the chemical; orconsent to future importation of the chemical subject to certain specified conditions.An interim response may include:
an interim decision either consenting to import with or without specified conditions, or not consenting to import during the interim period until a final decision is reached; ora statement that a final decision is under active consideration; ora request to the Secretariat, or to the Party that notified the final regulatory action, for further information; ora request to the Secretariat for assistance in evaluating the chemical.An import response form has been developed to facilitate the transmission of the import decisions to the Secretariat. The form with instructions on how to complete it may be found in Annex 5 to this guide or downloaded from the Rotterdam Convention web site www.pic.int
Points to consider in taking a decision
Parties need to be aware of the related provisions of the Convention in making an import response.
1. Category under which the chemical is included in the Convention
An import response shall relate to the category specified in Annex III of the Convention and in the DGD for the chemical.
For example:
aldrin is listed under the category pesticide, which includes all existing formulations subject to the PIC procedureimport decisions apply to all forms of aldrin used as a pesticide;phosphamidon is listed under the category severely hazardous pesticide formulationimport decisions apply specifically to soluble liquid formulations of phosphamidon that exceed 1000g active ingredient/limport decisions would only apply to the specific formulations identified;dinoseb is listed under the category pesticide, even though it also has uses as an industrial chemicalimport decisions would only apply to the use of dinoseb as a pesticide; imports for industrial chemical uses would not be included in the scope of the PIC procedure for this chemicalan importing Party may extend coverage of their import decision to the other category; however, there is no obligation under the Rotterdam Convention for the exporting Party for the chemicals for in the other category.2. Statement on legislation or administrative measures for a final import decision
A final decision on import should be accompanied by a description of any legislative or administrative measures upon which it is based. It could be any official document that further explains or implements the regulatory or import status of the chemical in the country, or an official document that deals with chemical regulation generally. This does not necessarily mean that legislative or administrative measures specifically to address the chemicals subject to the PIC procedure are required. For example, chemicals that have never been registered or approved for use in a country may be subject to a general regulation that does not allow their use or importation. Where information on legislative or administrative measures that are the basis for the final decision is provided by a Party, this is reflected in the PIC Circular together with the import decision.
3. Specifying conditions
As noted above, a final decision on import shall consist of one of three options: consent to import; no consent to import; or consent only subject to specified conditions. If the third option is selected, then the conditions for import need to be "specified". In other words, this suggests the need to state specifically the conditions under which export should be allowed. It might be considered insufficient, for example, simply to note that general conditions apply, or to fail to identify the specific conditions. Where such conditions are given in an import response, it is reflected in the PIC Circular together with the import decision.
4. Registration under national laws
Many Parties require that pesticides are not imported or used unless registered or authorized under national law. Some Parties describe this as a "no consent" situation, while others describe it as a "consent subject to conditions" situation. These different response patterns could create unintended confusion. Subject to any further guidance by Parties, and consistent with 2) and 3) above, references to national registration systems in import responses should be accompanied by sufficient information to clarify how the system applies to the chemical under consideration. One approach used by some Parties in this situation is to transmit a "no consent" response for chemicals that are not registered. It is then further explained (as applicable) that in the absence of official registration, a chemical may not be used or imported, and that registration may only be granted if a chemical complies with strict norms and a thorough regulatory review process. Where applicable, some Parties have also identified relevant exemptions, such as for purposes of research subject to specific conditions.
5. Trade neutrality
If the decision is not to consent to import or to consent to import only under specified conditions, this decision must apply equally to import of the chemical from any source and to domestic production of the chemical for domestic use. It is important to note that "any source" applies to both Parties and non-Parties.
Under the PIC procedure, exporting Parties must ensure that their exports of chemicals included in Annex III of the Convention are in line with the import decision of the importing Party. However, it should be noted that export of the chemicals listed in Annex III from exporting countries that are not Parties to the Convention may still take place contrary to the import decision of a Party, as non-Parties are not bound by the Convention. Importing Parties should endeavour to strengthen their own regulatory infrastructure as called upon by the Convention in Article 4, paragraph 2 and Article 15, paragraph 1.
How to reach to an import decisionA chemical is included in Annex III of the Convention because one or more Parties have determined that continued use of the chemical poses unacceptable risks and that use should cease (or be severely restricted). In the case of a severely hazardous pesticide formulation, it has been included because it has been shown that the pesticide formulation has caused a human health or environmental problems in a developing country or a country with an economy in transition under conditions of use.
The decision guidance document provides basic information about the chemical and the reasons for its listing in Annex III. It is intended to initiate an informed decision making process regarding future imports of the chemical in question. The decision guidance document can serve as a starting point for Parties in making a decision about a particular chemical, taking into account their national circumstances (see Figure 2). In addition, it is important that there be appropriate consultation at the national level with others involved within and outside the government, such as the Customs authority, trade authority and industry.
Parties need to assess whether the continued importation and use of the chemical poses an acceptable risk to human health or the environment, or if it can reasonably be managed within their chemical management infrastructure.
In addition to the decision guidance document, Parties may request further information from the Secretariat and notifying Parties. If additional technical information is requested, it should be clearly identified what type of information, such as more detailed information on ecotoxicity, characteristics of the chemical, etc. Parties may also seek assistance from the Secretariat in reaching a decision. Where such assistance is requested, the specific areas of difficulty and the nature of the assistance needed should be clearly indicated. An important element of reaching an import decision is whether or not there is domestic production of the chemical for domestic use. In this case, the DNA needs to have some level of information about chemical producers, products and usage in their country.
(Figure 2. The PIC procedure - national decision making process)
3.3.4 Obligations and procedures related to exports of chemicals included in Annex III
Obligations under Article 11 of the Convention
Article 11 sets out the obligations of Parties in relation to exports of chemicals included in Annex III of the Convention. These obligations can be implemented in a variety of ways. It is important to note that the Convention does not prescribe how Parties implement these obligations. Rather, governments must individually decide how they will ensure that exports do not occur contrary to importing Party decisions.
A further obligation for an exporting Party is to put in place legislative and/or administrative mechanisms that ensure that exporters do not export chemicals to importing Parties contrary to their import decisions. This mechanism is to be applied no later than six months after the Secretariat informs all Parties of the importing Party's decision in the relevant PIC Circular. The mechanism that governments choose to implement this obligation will depend upon their own legislative and administrative arrangements and available resources.
Parties are required to inform those concerned within their own territories of the import decisions of other Parties for chemicals included in Annex III of the Convention. This is to ensure that those involved with exports, or controls of exports, have the necessary information to ensure that exports do not take place contrary to decisions of importing Parties. Depending upon arrangements within the exporting Party, those concerned are likely to be chemical manufacturers, formulators and exporters and may include other government agencies involved in the regulation of chemicals, such as Customs or other border control agencies.
Process for the implementation of export responsibilities
The mechanism that governments choose to relay information on exports of Annex III chemicals and decisions of importing Parties to relevant stakeholders may include direct correspondence, the use of government publications such as gazettes or websites, the holding of seminars and workshops and advertisements or notices within the electronic or print media.
A possible mechanism could be to pass laws that make it an offence to export chemicals listed in Annex III of the Convention unless issued with a permit to do so. This would allow permits to be issued for Parties that have consented to further imports. This mechanism would require some level of border control for it to be effective, either through a specific service designed for such exports or through the utilization of existing systems such as a country's Customs or phytosanitary/quarantine controls.
Alternatively, Parties may elect to inform their industries of their responsibilities and require industry to self-regulate in compliance with this requirement. For this mechanism to be effective, it is likely that governments would still need to audit exportation and have some means of enforcing obligations where industry has been found not to have complied.
An important aspect of obligations regarding chemicals listed in Annex III of the Convention is that the export obligations only apply to exports of the chemical where it is destined for use in the category for which it was included in the Convention (i.e. pesticide, industrial chemical or SHPF).
Annex III of the Convention indicates the category for which a chemical has been listed. Most chemicals clearly fall into one category (i.e. most pesticides do not have an industrial chemical use and most industrial chemicals are not used as pesticides). However, there are a small number of chemicals with uses as both a pesticide and an industrial chemical, or so-called "dual use" chemicals. Where such chemicals are included in the Convention under a single category, then there are no obligations on the exporting Party if the chemical is being exported for use in the importing Party in the other category.
The difficulty for DNAs in exporting Parties is in knowing, at the point of export, what the intended use is to be in the importing country. For example:
if the chemical being exported is already pre-formulated and labelled for pesticide use, then this would provide a very strong indication of its intended use. However, most trade in international chemicals occurs in bulk form and this would not, in general, enable a determination to be made as to its intended use (or in the case of an active ingredient for a SHPF, what type and concentration of formulation it might become). Exporting Party DNAs might consider some means to ensure import is compliant. One way of doing this is by requiring a documented declaration from the importing agent, through the exporter, as to its intended use before deciding whether to allow the export.Responsibility of an Exporting Party when there is no import response or an interim response without a decision
On occasion, a Party will fail to send an import response or will send an interim import response that does not contain an import decision. These failures to transmit a response are listed in Appendix IV of the PIC.
In such circumstancies, an exporting Party must still ensure that exportation of an Annex III chemical does not occur, unless:
the chemical is registered in the importing Party at the time of export; orthe exporting Party has evidence or information that the chemical has been previously imported or used in the importing Party and that there has been no consequent prohibition of that use or importation; orthe DNA of the importing Party has provided explicit consent to the exporter.This obligation of exporting Parties not to export an Annex III chemical unless one of the above conditions is met only applies for one year it commences six months after the date when the Secretariat informed Parties that the country had not sent an import response or had sent an interim response that did not contain a decision regarding import. This obligations, however, regarding export of an Annex III chemical only applies for one year and therefore ceases 18 months after the date when the Secretariat informed Parties of the failure to provide a decision.
Information to Accompany Exports
Article 13 outlines the information to accompany exports of chemicals included in Annex III of the Convention as well as chemicals that are banned or severely restricted in the exporting Party. Such exports should have labelling that provides adequate information on the hazards and risks posed by the chemical to human health and the environment. The Convention also requires that for such exports a copy of an up-to-date Safety Data Sheet (SDS), in an internationally recognized format, be sent to the importer. Furthermore, the information on the label and the safety data sheet should be in the official language of the importing country, as far as is practicable.
The Convention also requires that shipping documents for exports of chemicals listed in Annex III should bear the specific Harmonized System Customs code (HS codes) assigned by the World Customs Organization (WCO). Annex 4 of this guidance document contains a list of the HS codes which have been assigned by the WCO to the chemicals included in Annex III.
The purpose of these requirements is to ensure that for certain hazardous chemicals there is sufficient information provided on labels and in the SDS to minimize risks to workers, others and the environment.
3.4 How the Prior Informed Consent (PIC) Procedure benefits Parties
Benefits to Parties include:
Early warning / Information Exchange
Parties are made aware of national bans or severe restrictions of hazardous chemicals in other Parties through publication of the summaries of notifications of final regulatory action in Appendix IV of the PIC Circular. This early warning of potentially hazardous chemicals is a source of information on why countries have taken domestic regulatory actions and may provide an incentive for other countries, particularly countries that import such chemicals, to review the regulatory status of these chemicals. In addition, the PIC Circular also contains summary reports on human poisonings and environmental damage associated with the use of severely hazardous pesticide formulations. This information may alert countries to possible problems that may be occurring in their own territory.
Informed decision–making
Parties receive decision guidance documents for each of the chemicals included in Annex III of the Convention. The information contained in the decision guidance document provides the basis for initiating a process for making informed decisions regarding future imports of these chemicals.
Shared responsibility
The PIC procedure helps Parties to better manage chemicals by promoting a shared responsibility among exporting countries, importing countries and industry. Importing Parties have a responsibility to make informed decisions regarding future import in a timely manner, while exporting Parties have a responsibility to ensure that exports do not occur contrary to the decisions of importing Parties. In participating in the PIC procedure, Parties should recognize that non-Parties to the Convention may still export chemicals listed in Annex III of the Convention.
Networks among Parties
As a Party to the Rotterdam Convention, designated national authorities (DNAs) form part of a global network of government officials involved in chemicals management. This should facilitate an exchange of ideas and create opportunities for cooperative efforts to resolve issues that could not be easily resolved by one country alone.
THE PRIOR INFORMED CONSENT (PIC) PROCEDURE
EXPORT NOTIFICATION AND INFORMATION TO ACCOMPANY EXPORT
4. Export notification and information to
accompany export
4.1 Introduction
The Convention, in Articles 12 and 13, sets out certain requirements to ensure that adequate information is made available to importing Parties when export occurs of a chemical in Annex III or a chemical that has been banned or severely restricted in the exporting Party.
This section summarizes the key provisions of the Convention concerning export notification and information to accompany export and the key tools and mechanisms that have been developed to implement those provisions.
Article 12 outlines the obligations and the process for export notification. Export notification is a mechanism to promote the exchange of information among countries regarding banned and severely restricted chemicals. The process alerts Parties that they are receiving imports of a chemical that has been banned or severely restricted in the exporting Party.
Export notification is different from the PIC procedure in that it does not ask Parties for a decision regarding future imports of the chemical. It simply informs that a shipment of a chemical that has been banned or severely restricted in the exporting Party is being sent.Article 13 outlines the information that is to accompany shipments of exported chemicals, both those that are included in Annex III to the Convention and those that are banned or severely restricted in the exporting Party. The purpose of this provision is to ensure that the identity of and information on the chemical are provided to importing Parties to assist them in minimizing risks to human health and the environment from such chemicals.
4.2 How the Export Notification Process Works
The Convention requires a Party to send an export notification to an importing Party prior to the first export in a calendar year of a chemical that is banned or severely restricted in the exporting Party's territory. The exporting Party is also required to provide an updated export notification if it has adopted a final regulatory action that makes a major change in its previous regulatory decision. An example of such a change might be where the chemical was originally severely restricted and subsequently the exporting country took further action to ban the chemical.
An importing Party is required to acknowledge receipt of the export notification to the exporting Party. If no such acknowledgement is received by the exporting Party after 30 days, the exporting Party is required to send a second notification and make reasonable efforts to ensure that the importing Party receives the notification.
The obligation for an exporting Party to send export notifications to an importing Party ends when the chemical is included in Annex III to the Convention, the importing Party has provided an import response for the chemical and the Secretariat has circulated import responses for that chemical to all Parties in the PIC Circular.
It is important to note that, unlike Article 11 on the obligations of Parties concerning exports of chemicals subject to the PIC procedure, Article 12 on export notifications does not specify differential obligations regarding the category of chemical for which the final regulatory action was taken. As a result, Parties have interpreted their obligation for export notifications in a variety of ways:
Some Parties have interpreted their obligations such that they provide an export notification for a chemical that is banned or severely restricted domestically in one category irrespective of what the intended use category will be for the chemical in the importing Party.Others Parties have interpreted their obligations such that they will only provide an export notification when the intended use in the importing Party is in the same use category as that for which their final regulatory action was taken.Parties should make their own decision as to how they will interpret and implement export notification with regard to this matter.
The Secretariat has developed a standard form for export notification in order to facilitate Parties in meeting their obligations under Article 12. Parties are encouraged to use this form when making or acknowledging receipt of export notifications. Forms that have been developed at the national level that meet the information requirements of Annex V of the Convention may continue to be used (Annex 5).
4.3 How Export Notification Benefits Parties
Parties are first informed about a potentially hazardous chemical when a summary of a notification of final regulatory action to ban or severely restrict its use in a Party is published in Appendix I of the PIC Circular.
Export notifications thus serve as an individual reminder to importing Parties of a final regulatory action taken by the exporting Party. An export notification alerts the importing Party that a chemical banned for human health or environmental reasons in another Party may be in use in their country. The importing Party may take the opportunity to seek further information on the chemical from the exporting Party or from other sources and to consider whether it might need to apply any risk management actions regarding the chemical concerned.
4.4 Information to accompany Exports
Article 13 outlines the information that is to accompany exported chemicals, both those that are included in Annex III to the Convention and those that are banned or severely restricted in the exporting Party. The purpose of this provision is to ensure that information is provided to importing Parties to assist them in minimizing risks to human health and the environment from such chemicals.
The specific requirements include:
specific Harmonized System Customs codes for chemicals in Annex III assigned by the World Customs Organization;labelling that provides adequate information on the hazards and risks posed by the chemical to human health and the environment;a copy of an up-to-date Safety Data Sheet (SDS) in an internationally recognized format is to be sent to the importer. The information on the label and the SDS should be in the official language of the importing country as far as is practicable.The World Customs Organization has assigned specific Harmonized System Customs codes to the individual chemicals or groups of chemicals listed in Annex III (see Annex 6 of this Guide). These codes entered into force on 1 January 2007. The Convention, in Article 13, requires that whenever a code has been assigned to a chemical in Annex III, the shipping document for that chemical has to bear the code when exported.
Exporting Parties are also to ensure that chemicals when exported are subject to environmental and health labelling requirements in their territories are, when exported, subject to labelling requirements that ensure adequate availability of information with regard to risks and/or hazards to human health and the environment.
4.5 How Information to Accompany Exports Benefits Parties
The Harmonized Systems Customs codes (HS codes) provide an international standard for the identity of the chemicals listed in Annex III. The specific HS codes for chemicals in Annex III of the Convention help facilitate integration of work on the Rotterdam Convention with the current activities of Customs authorities and contribute to the effective implementation of the PIC procedure.
Inappropriate use of hazardous chemicals is often due to a lack of correct information, e.g. label in a foreign language. The Convention obliges exporting Parties to provide adequate labelling and a safety data sheet to each importer, according to international standards and in the official language(s) of the importing Parties as far as practicable. This is to ensure the adequate availability of information with regard to risk or hazard of the chemical. With this information the Convention assists governments in minimizing the impact of hazardous pesticides and chemicals to human health and the environment.
5. Information exchange
5.1 Introduction
Information exchange is an integral part of the Rotterdam Convention. The opportunities for information exchange cover a broad range of chemicals, those included in Annex III of the Convention as well as chemicals that have been banned or severely restricted in any Party. In addition, Article 14 mandates a general exchange of scientific or regulatory information “relevant to the objectives of the Convention” of potential interest to regulators around the world.
This section summarizes the key provisions of the Convention concerning information exchange and the key tools and mechanisms that have been developed to implement those provisions.
5.2 How Information Exchange Works
The various provisions of the Convention provide opportunities to obtain information on potentially hazardous chemicals and to share information and experience with countries facing similar concerns. The most important tools for information exchange include the PIC Circular, export notifications, decision guidance documents, the network of DNAs as well as the Rotterdam Convention web site.
5.2.1 Key provisions of information exchange
The key provisions of information exchange under the Rotterdam Convention may be summarized as follows:
Notifications of final regulatory action (see Section 2.3.1)
If a Party takes a final regulatory action to ban or severely restrict a chemical for health or environment reasons, it shares the information with all Parties by notifying the action to the Secretariat. Summaries of each notification received by the Secretariat are published in the PIC Circular and sent to all DNAs. The summaries briefly describe the scope of the action and the reasons for it, including information on hazards and risks of the chemical to human health or the environment, and the expected effect of the regulatory action.
Proposals for severely hazardous pesticide formulations (see Section 2.3.2)
If a developing country Party or Party with an economy in transition experiences problems with a certain pesticide formulation under the conditions of use in its territory, it can share the information with all Parties by proposing the inclusion of the formulation in Annex III to the Secretariat. Summaries of each proposal for inclusion of an SHPF received by the Secretariat are published in the PIC Circular and sent to all DNAs. The summaries briefly describe the incident, the adverse effect on human health or the environment and how the formulation was used.
Export notifications (see Section 4.2)
When a Party is going to export a chemical that is banned or severely restricted in its own territory, it is required to send an export notification to the importing Party prior to the first export of that chemical in a calendar year. Export notification helps to make the importing Party aware of the fact that they are going to receive a chemical that is banned or severely restricted in the exporting Party. Export notification provides the importing Party with relevant information for identifying the chemical and the shipment as well as information related to its known risks and hazards.
Information to accompany exports (see section 4.4)
When a Party exports chemicals, both those that are included in Annex III to the Convention and those that are banned or severely restricted domestically, it has to provide certain information with the shipment. This information includes labelling and safety data sheets according to international standards and, as far as practicable, information must be in the official languages of the importing Party.
The Convention further requires that where a specific Harmonized System Customs code has been assigned to a chemical in Annex III, the shipping documents for that chemical include this code.
Exchange of general information on chemicals
In line with the objectives of the Convention, Parties are encouraged to share general information concerning chemicals within the scope of the Convention. Article 14 of the Convention specifically addresses information exchange and requires Parties to facilitate the exchange of scientific, technical, economic and legal information. This includes providing information that is publicly available on their regulatory actions and also making this available to other Parties either directly or through the Secretariat.
The Convention obliges Parties to protect confidential information. However, it specifies certain types of information that are not to be regarded as confidential. This information includes, among others, the information provided as part of notifications of final regulatory action or proposals for hazardous pesticide formulations to the Secretariat, the expiry date of the chemicals and summaries of toxicological and eco-toxicological tests.
If a Party requests the Secretariat to facilitate the exchange of information as described above then this is done through the PIC Circular and the Convention web site www.pic.int.
5.2.2 Key tools of information exchange
The key tools to facilitate the exchange of information under the Convention include the following:
PIC Circular
The PIC Circular (see Section 1.4.3 and Annex 2) contains information on national final regulatory actions to ban or severely restrict chemicals as well as proposals for hazardous pesticide formulations submitted by Parties. It also includes an up-to-date list of the chemicals subject to the PIC procedure, a compilation of all the import decisions of Parties as well as those Parties that have failed to provide an import decision.
Should a Party wish to share information on domestic regulatory actions through the Secretariat, or require information on transit movements of Annex III chemicals through its territory, this may be published in the PIC Circular.
The PIC Circular is published by the Secretariat twice a year, in June and December, sent to all DNAs and posted on the Convention web site.
Decision Guidance Documents
Parties receive information on the chemicals subject to the PIC procedure through each chemical's decision guidance document (see Section 1.4.3 and Annex 1). These documents contain a summary of important information related to the final regulatory actions taken by notifying Parties, basic information about the characteristics of the chemical and summary information about the hazards and risks associated with the use of the chemical. Decision guidance documents are sent to DNAs by the Secretariat when a chemical is added to Annex III and are available on the Convention web site www.pic.int.
Contact information for Designated National Authorities
The Secretariat maintains a list of the contact details for all designated national authorities (DNAs) of Parties to the Convention. This list provide for easy provides access to a network for the exchange of information on chemicals of concern. A cCurrent version of the list of contact details for DNAs is available on the Convention website www.pic.int and is also circulated to all DNAs by the Secretariat together with the PIC Circular every six months and posted on the Convention web site www.pic.int.
Rotterdam Convention Web site www.pic.int
The Rotterdam Convention web site is an important source of information on the operation and status of implementation of the Convention. It provides a current list of Parties to the Convention and the contact details of DNAs. It also provides access to a database of all import responses for Annex III chemicals submitted by Parties, and a database listing all notifications of final regulatory actions received from Parties. Documents including the PIC Circular, decision guidance documents, and relevant forms and instructions are available on the web site. The web site is updated on an ongoing basis to provide Parties with up-to-date and timely information.
Highlights of other information found on the website include:
Additional information on Annex III chemicalsInformation on additional notifications of final regulatory actions to ban or severely restrict Annex III chemicals as well as information on alternatives, national risk evaluations and evaluations from internationally peer reviewed processes such as those of the World Health Organization (WHO), the International Agency for Research on Cancer (IARC) and safety data sheets are also provided in the website.
COP and CRC meetings
Information on upcoming meetings of the Conference of the Parties and Chemical Review Committee, and the associated meeting documents, are posted in advance of the meetings, and reports are posted subsequently.
Technical assistance
The web site provides information on the different types of technical assistance activities facilitated by the Secretariat, as well as a schedule of upcoming technical assistance activities, countries involved and reports of past activities.
Country profiles
Each Party to the Convention has a "country profile" page, which contains all information on the status of implementation of the Convention for that Party, including date of ratification, contact details for Official Contact Points and Designated National Authorities, import responses for Annex III chemicals and final regulatory action notifications. It also lists all of the technical assistance activities in which the country has participated.
Resource kit
The Resource Kit, a collection of documents and publications that provide detailed information on various aspects of the Convention, is also available. These publications are generally available for download from the website in English, French, Spanish, Arabic, Chinese and Russian.
5.3 How Information Exchange Benefits Parties
The information exchange provisions of the Rotterdam Convention build networks of persons that individual DNAs may call upon At the sub-regional, regional and global level this can create a pool of expertise that can enable an exchange of ideas and possible cooperative resolution of problems that may not be achievable by a country acting alone. The receipt of additional information that may not otherwise be available to a country provides a broader base upon which sound regulatory decisions may be made, delivering better health and environmental outcomes.
Information exchange also has a direct effect on resource allocation. If information on certain chemicals is available from other Parties, the Secretariat or international sources, then Parties may not need to use up resources in trying to collect this information. The often limited resources that are available to some Parties may then instead be allocated to other aspects of chemical management.
Depending upon the characteristics of the chemical concerned, information exchanged under the Rotterdam Convention may also help to highlight chemicals that are persistent, bio-accumulative and toxic and therefore possibly eligible for consideration in other fora, such as the Stockholm Convention on Persistent Organic Pollutants (POPs).
6. Responsibilities of DNAs and governments in implementing the Rotterdam Convention
Under the Convention, Parties have certain obligations which often help to increase the effectiveness of the Convention. The following is a checklist of actions that Governments/DNAs must have performed for a country to fully participate in the Rotterdam Convention.
Yes No
Has your government nominated a DNA for both pesticides and industrial chemicals?
Have the DNAs been provided with sufficient resources to carry out their duties?
Has the DNA(s) sent completed notification forms for chemicals that have been banned or severely restricted in your country to the Secretariat?
Has the DNA(s) sent completed import response forms containing a decision on future imports for all chemicals listed in Annex III of the Convention to the Secretariat?
Has domestic production for domestic use or importation from non-Party sources been prohibited for any chemical listed in Annex III of the Convention for which your country's import decision was to not allow future import?
Have the import decisions of other Parties been communicated to other relevant agencies and industries in your country?
Does a mechanism exist in your country to ensure that there will be no exports of chemicals in Annex III to the Convention to Parties that do not wish to receive them?
Does a mechanism(s) exist in your country for export notifications to be sent with the first export in each calendar year of domestically banned or severely restricted chemicals that are not yet included in Annex III of the Convention?
Do you have procedures in place to enable you to promptly provide a notice of receipt regarding any export notifications received?
Are there mechanism(s) in place to ensure that exports of chemical are accompanied by the necessary information required under the Convention?
For developing countries and countries with economies in transition, do you have mechanisms in place that will enable you to identify proposals for severely hazardous pesticide formulations?
6.1 Getting Started
Under Article 4 of the Convention, Parties are to designate one or more national authorities to act on their behalf with regard to the performance of the administrative functions required by the Convention. Designated National Authorities (DNAs) are the key point of contact between a Party and the Secretariat and between Parties in the implementation of the Convention, in particular concerning the PIC procedure and export notification provisions of the Convention. DNAs are also responsible for ensuring the dissemination of information concerning the Rotterdam Convention at the national level, including to other Ministries with a role in chemicals management, such as Customs authorities, exporting and importing industries and other stakeholders.
Given the technical nature of the responsibilities of a DNA, most Parties have nominated a government ministry or agency that is actively involved in regulating chemicals or pesticides in their country with the understanding that the work on the Rotterdam Convention may be integrated with their existing activities. In view of the frequency with which there is a change of personnel in many countries, it is generally preferable that an office or a particular position, rather than a particular person, is identified as the DNA (e.g. the office of the pesticide registrar or the pesticide registration board). In some instances, DNAs have been nominated from ministries of foreign affairs or at the ministerial level. While in some countries these ministries may play a coordinating role in chemicals management activities at the national level, it is difficult to assess the ability of these ministries or senior officials to fulfil the day to day responsibilities of the DNA.
The decision as to how many DNAs are nominated is up to individual governments. Many Parties have nominated only one while others have nominated two. The nomination of DNAs generally reflects the domestic arrangements for chemicals management, with one DNA being responsible for industrial chemicals and a second for pesticides. In some cases, Parties have nominated one DNA for both industrial chemicals and pesticides while others have nominated only one DNA for one category of chemicals; for example, pesticides. In such situations, where there is no nominated DNA for the other category, governments should examine their situation to ensure that they have adequate arrangements in place to fully participate in the Rotterdam Convention. If a country has more than one DNA, they need to ensure that they coordinate and establish clear areas of responsibility to ensure submissions to the Secretariat are complementary rather than contradictory.
The nomination or change of nomination for a DNA should be sent from the country’s Official Contact Pointfor the Rotterdam Convention to the Secretariat. Similarly, any change in contact information for the DNA should be submitted to the Secretariat. Changes in address or other contact information within an established DNA office may be submitted directly by the DNA. Changes in institution of an established DNA would be considered a new nomination and should therefore be submitted through the Official Contact Point. The Secretariat maintains the list contact information of DNAs which is posted on the Convention website and circulated to all Parties every six months together with the PIC circular.
6.2 Responsibilities of the Designated National Authority (DNA)
The DNA for a Party undertakes a number of responsibilities which are described in Articles 5, 6, 10 and 12. These include:
Notifying the Secretariat of national final regulatory actions (see Section 2.3.1)
Under Article 5 of the Convention, Parties have the following obligations with respect to notifying the Secretariat of their final regulatory actions to ban or severely restrict a chemical for health or environmental reasons:Submitting proposals for severely hazardous pesticide formulations (SHPFs) (see Section 2.3.2)
Under Article 6 of the Convention, DNAs from developing country Parties and Parties with an economy in transition may submit proposals to the Secretariat for the listing of SHPFs in Annex III of the Convention, if they are experiencing problems caused by such formulations under conditions of use in their countries. In developing these proposals they may draw upon technical expertise from any relevant source.Providing import responses to chemicals subject to the PIC procedure (chemicals in Annex III of the Convention) (see Section 3.3.3)
Under Article 10 of the Convention, Parties have the following obligations with respect to imports of chemicals listed in Annex III of the Convention and subject to the PIC procedure:Communicating import responses to stakeholders in a country
Designated National Authorities receive a complete set of all import responses every six months through the PIC Circular. DNAs are expected to disseminate this information to all relevant agencies that may be involved in the regulation, production and trade of chemicals in their country (e.g. government departments, manufacturers, export industries, etc.). This is to enable those institutions and persons to take the appropriate action to ensure that exports do not occur contrary to the import decisions of Parties.6.3 Other obligations for Parties
Apart from the above-mentioned obligations, governments should consider what other actions have to be taken from their side in order to fully participate in and apply the Convention.
Parties must equally apply their import decision to all sources and to the domestic manufacture of the chemical for domestic use
When a Party decides not to allow the future import of a chemical listed in Annex III of the Convention or to allow import subject to certain conditions, then that Party must also prohibit or apply the same conditions to the import of the same chemical from all sources. These sources will include trade from non-Parties to the Convention and when the chemical is not imported but is produced domestically for domestic use. This will require Parties to have legislative or administrative mechanisms in place to prohibit import at its borders and to prevent manufacture of the chemical within its own territory.
Many countries may have pesticide registration schemes covering the registration of pesticides and pesticide formulations, but not all countries have equivalent schemes that can effectively regulate both the import and the manufacture of industrial chemicals. Therefore, there may be a need for some countries to strengthen their domestic regulatory schemes.
Most countries also have established Customs and quarantine schemes, which are enforced at their borders, and these may provide a mechanism that could (with appropriate amendments) be used to control the import (and export) of the relevant chemicals. Other mechanisms may also exist depending upon a country’s legislative and administrative infrastructure.
Parties must ensure that export does not occur contrary to import decisions
One of the most important elements of the Convention that Parties must implement is the ability to ensure that exports of chemicals listed in Annex III of the Convention do not occur contrary to the decisions of importing Parties.
Parties therefore need to ensure that either legislative or administrative mechanisms are in place that prohibit the exports of these chemicals. One possible mechanism is to use existing Customs legislation and border controls. Other possible mechanisms may be:
to impose a prohibition on the export of Annex III chemicals and require industry to comply;to establish a licensing system for exporters of chemicals, with non-compliance in regard to the export of chemicals in Annex III of the Convention leading to possible cancellation of licences;to establish an education programme directed at raising awareness of the responsibilities under the Rotterdam Convention and PIC procedure among the chemical and exporting industries; and/orother mechanisms consistent with the exporting Party’s legislative and administrative infrastructure.Whichever mechanism is chosen, it is important to recognize that there will need to be resources devoted to inspection and enforcement to ensure that compliance is achieved.
Parties must ensure human health and environment information accompany exports
The obligations that human health and environment information accompany the export of chemicals will require some Parties to strengthen their chemicals management system. In many countries, the existing labelling and other requirements regarding information on chemicals may ensure that any chemical destined for export already has adequate information accompanying the shipment. If this is not the case, Parties will need to ensure, through either education campaigns or formal legislative provisions, that exports of chemicals are accompanied by adequate information regarding protection of health and the environment, as required by the Convention.
6.4 Technical Assistance
Article 16 of the Convention contains a general obligation for Parties to recognize the needs of developing country Parties and Parties with economies in transition and to provide technical assistance to build capacity for chemicals management and to enable implementation of the Convention.
The Rotterdam Convention has been developed to take into account the needs of Parties with regard to economic conditions, legislation, awareness, knowledge of decision makers and access to information. Therefore, the Convention requires that Parties with more advanced programmes should provide technical assistance, including training, to other Parties in developing their infrastructure and capacity to manage chemicals, in order to enable all Parties to fully implement the Convention.
A programme of technical assistance is discussed and agreed at each meeting of the Conference of the Parties (COP). A voluntary trust fund has been established to support the programme. The Secretariat has been mandated by the COP to facilitate the regional and national delivery of technical assistance.
Detailed information on technical assistance activities can be found on the Convention web site www.pic.int.
RESPONSIBILITIES OF DNAs AND GOVERNMENTS IN IMPLEMENTING THE ROTTERDAM CONVENTION
6 Checklist of actions
7. Coordination with key stakeholders at
national level
A key function o f the Rotterdam Convention is to help countries strengthen their chemicals management infrastructure and take informed decisions aimed at preventing unwanted trade in certain hazardous chemicals and pesticides. The effective implementation of the Convention at the national level requires adequate information exchange and coordination with key stakeholders both within government as well as outside government. As a first step Parties need to ensure that adequate mechanisms are in place so that the coordination between the Designated National Authority (DNA) and key stakeholders occurs. Recognizing that different countries may apply different processes, this section provides some points that might be considered in working with key stakeholders.
7.1 Government stakeholders
There are a broad range of stakeholders within government that would be expected to work with the DNA in implementing the various provisions of the Convention. DNAs, in coordination with government stakeholders, will need to ensure that there is an appropriate legislative and administrative basis for the effective implementation and enforcement of the Convention.
The information on banned or severely restricted chemicals available through the information exchange provisions of the Convention would be expected to be useful to those ministries or authorities actively engaged in the regulation of chemicals and pesticides. These authorities would be likely to include, although may not be limited to, ministries of agriculture, environment and health. A mechanism to ensure the timely circulation of information at the national level, as well as integrating such information with the chemicals review process, should be considered. Such authorities may also be involved in ensuring that information relating to notifications of final regulatory action is compiled and submitted to the Secretariat.
In submitting a proposal relating to a severely hazardous pesticide formulation, input may be sought by the DNA from relevant areas of health or agriculture. Regional health clinics and agricultural extension personnel might also be able to provide information relating to problems with hazardous pesticide formulations under the conditions of use in the country.
The implementation of the PIC procedure as well as provisions concerning labelling and the information to accompany exported chemicals might be expected to involve interaction with a broader range of ministries including ministries of trade or commerce and foreign affairs, as well as Customs authorities. In taking an import decision, consultations may involve all of these stakeholders, and may also require input from non-government stakeholders, In implementing the PIC procedure DNAs and responsible government agencies should establish mechanisms to ensure timely dissemination of all import responses for Annex III chemicals, including their own domestic import decisions.
Where there is clear domestic regulation of the use of chemicals, the responsible government agencies, such as the pesticide or industrial chemical authority responsible for registration, may have established lines of communication with domestic manufacturers and formulators. Such links could play a key role in the implementation of the PIC procedure by helping to ensure that, when a national import decision is not to consent to import, there is no domestic production for domestic use of that chemical.
Similarly, Customs authorities have a role to play in ensuring that any exports of Annex III chemicals reflect the decisions of importing countries and that imports do not occur contrary to import decisions.
Customs authorities also have a possible role in ensuring that, when exported, chemicals in Annex III or those that are banned or severely restricted domestically, are in compliance with the provisions of the Convention concerning labelling and information to accompany shipments of such chemicals (see Section 3.3.3 and 3.3.4 of this guide).
7.2 Elements relevant to the Customs officials
Effective 1 January 2007, specific Harmonized System Customs codes for most of the chemicals in Annex III have been assigned by the World Customs Organization (WCO) (see Annex 4). These codes replace the previous codes for some Annex III chemicals which related only to either their chemical structure or the proposed use of the product, rather than to their inclusion in the Rotterdam Convention. The inclusion of these codes in the shipping documentation for these chemicals, as required by the Convention, should facilitate integration of the work on the Rotterdam Convention with that of Customs authorities. As many countries are already members of the WCO, most Customs authorities are already familiar with the use of these codes.
To assist with the implementation of the Rotterdam Convention, DNAs and Customs officials should develop an enforcement procedure in support of the implementation of the relevant provisions of the Convention. A list of the key elements that might be considered in developing such a procedure relevant to the PIC procedure and the provisions of the Convention concerning export notification and the information to accompany exported chemicals includes the following:
PIC procedure:
ensure a current list of chemicals in Annex III is availableintegrate the specific HS codes for chemicals in Annex III in the domestic Customs systemensure that imports of chemicals in Annex III comply with national import decisions and that the import decisions are up-to-dateensure that exports of chemicals in Annex III comply with import decisions from all other Parties and that the import decisions are up-to-date.Provisions related to export notification and the information to accompany exported chemicals:
ensure that the list of chemicals banned or severely restricted domestically that are therefore subject to export notification is up-to-dateensure that the list of Annex III chemicals that are manufactured or formulated in the country and exported is up-to-dateimplement a procedure to check whether an export notification has been sent for the first export to a Party in the calendar yearfor chemicals in Annex III and those that are banned or severely restricted domestically, ensure that labelling provisions of the Convention are met and that the information is provided to accompany exports including Harmonized System Custom codes and safety data sheets.Countries have taken different approaches at national level to implement the provisions of the Convention for the PIC procedure and for the provisions concerning export notification and the information to accompany exported chemicals. One example has been to establish a permit system for the import or export of certain chemicals, e.g. those chemicals in Annex III, as well as those chemicals banned or severely restricted in the country. In such cases, the permits may be issued by the DNA and included in the shipping documents that are to be checked by Customs officers. In doing so, DNAs must ensure that permits for the import or export of Annex III chemicals are only issued in compliance with the relevant import decisions.
Similarly, permits for the export of these chemicals are only issued upon confirmation that the provisions of the Convention, concerning export notification and the information to accompany exported chemicals such as labelling requirements or safety data sheet, have been met.
In such cases, there are three principal tasks for Customs officers:
identify the chemical in questionif the chemical falls within the scope of the Rotterdam Convention (PIC procedure or banned or severely restricted nationally) allow shipment only upon the issuance of a permit by the DNA; andin the absence of a permit from the DNA, Customs officers should stop the shipment and contact the DNA.7.3 Non-Governmental Stakeholders
The non-governmental stakeholders involved in the implementation of the Convention will vary among Parties. They might be considered to include associations of chemical manufacturers or formulators, commodity associations (including farmers associations or lobby groups), trade unions and environmental or consumer safety groups or other representatives of civil society with an interest or role in chemicals management.
The information available through the Convention such as improved labelling and the provision of safety data sheets with individual shipments of chemicals should help to reduce risks to human health and the environment from hazardous chemicals and should be made available to the relevant stakeholders.
In the implementation of the Convention it is also important to ensure that the national chemical industry, including manufacturers, formulators and traders, are aware of the provisions of the Convention and the possible impacts on their activities. In particular, with respect to the PIC procedure, DNAs should ensure they communicate to those stakeholders the national import decisions as well as those of other Parties. Chemical industry representatives should also be kept informed of developments in the Convention, in particular the listing of additional chemicals in Annex III. Such groups may also need to be consulted as part of the process of taking decisions on future import of chemicals.
The procedures for severely hazardous pesticide formulations set out in Article 6 clearly state that in developing a proposal for a severely hazardous pesticide formulation a Party may draw upon technical expertise from any relevant source. This could include groups or individuals involved in monitoring the health of workers that use pesticides, trade unions or others. Other sources of technical expertise may include environmental non-governmental organizations and farmers groups.
COORDINATION WITH KEY STAKEHOLDERS AT NATIONAL LEVEL
Contextual menu
Next
chapter
Previous chapter
Download text as PDF
7. Coordination with key stakeholders at
national level
A key function o f the Rotterdam Convention is to help countries strengthen their chemicals management infrastructure and take informed decisions aimed at preventing unwanted trade in certain hazardous chemicals and pesticides. The effective implementation of the Convention at the national level requires adequate information exchange and coordination with key stakeholders both within government as well as outside government. As a first step Parties need to ensure that adequate mechanisms are in place so that the coordination between the Designated National Authority (DNA) and key stakeholders occurs. Recognizing that different countries may apply different processes, this section provides some points that might be considered in working with key stakeholders.
7.1 Government stakeholders
There are a broad range of stakeholders within government that would be expected to work with the DNA in implementing the various provisions of the Convention. DNAs, in coordination with government stakeholders, will need to ensure that there is an appropriate legislative and administrative basis for the effective implementation and enforcement of the Convention.
The information on banned or severely restricted chemicals available through the information exchange provisions of the Convention would be expected to be useful to those ministries or authorities actively engaged in the regulation of chemicals and pesticides. These authorities would be likely to include, although may not be limited to, ministries of agriculture, environment and health. A mechanism to ensure the timely circulation of information at the national level, as well as integrating such information with the chemicals review process, should be considered. Such authorities may also be involved in ensuring that information relating to notifications of final regulatory action is compiled and submitted to the Secretariat.
In submitting a proposal relating to a severely hazardous pesticide formulation, input may be sought by the DNA from relevant areas of health or agriculture. Regional health clinics and agricultural extension personnel might also be able to provide information relating to problems with hazardous pesticide formulations under the conditions of use in the country.
The implementation of the PIC procedure as well as provisions concerning labelling and the information to accompany exported chemicals might be expected to involve interaction with a broader range of ministries including ministries of trade or commerce and foreign affairs, as well as Customs authorities. In taking an import decision, consultations may involve all of these stakeholders, and may also require input from non-government stakeholders, In implementing the PIC procedure DNAs and responsible government agencies should establish mechanisms to ensure timely dissemination of all import responses for Annex III chemicals, including their import decisions.
Where there is clear domestic regulation of the use of chemicals, the responsible government agencies, such as the pesticide or industrial chemical authority responsible for registration, may have established lines of communication with domestic manufacturers and formulators.
Such links could play a key role in the implementation of the PIC procedure by helping to ensure that, when a national import decision is not to consent to import, there is no domestic production for domestic use of that chemical.
Similarly, Customs authorities have a role to play in ensuring that any exports of Annex III chemicals reflect the decisions of importing countries and that imports do not occur contrary to import decisions.
Customs authorities also have a possible role in ensuring that, when exported, chemicals in Annex III or those that are banned or severely restricted domestically, are in compliance with the provisions of the Convention concerning labelling and information to accompany shipments of such chemicals (see Section 3.3.3 and 3.3.4 of this guide).
7.2 Elements relevant to the Customs officials
Effective 1 January 2007, specific Harmonized System Customs codes for most of the chemicals in Annex III have been assigned by the World Customs Organization (WCO) (see Annex 11). These codes replace the previous codes for some Annex III chemicals which related only to either their chemical structure or the proposed use of the product, rather than to their inclusion in the Rotterdam Convention. The inclusion of these codes in the shipping documentation for these chemicals, as required by the Convention, should facilitate integration of the work on the Rotterdam Convention with that of Customs authorities. As many countries are already members of the WCO, most Customs authorities are already familiar with the use of these codes.
To assist with the implementation of the Rotterdam Convention, DNAs and Customs officials should develop an enforcement procedure in support of the implementation of the relevant provisions of the Convention. A list of the key elements that might be considered in developing such a procedure relevant to the PIC procedure and the provisions of the Convention concerning export notification and the information to accompany exported chemicals includes the following:
PIC procedure:
ensure a current list of chemicals in Annex III is availableintegrate the specific HS codes for chemicals in Annex III in the domestic Customs systemensure that imports of chemicals in Annex III comply with national import decisions and that the import decisions are up-to-dateensure that exports of chemicals in Annex III comply with import decisions from all other Parties and that the import decisions are up-to-date.Provisions related to export notification and the information to accompany exported chemicals:
ensure that the list of chemicals banned or severely restricted domestically that are therefore subject to export notification is up-to-dateensure that the list of Annex III chemicals that are manufactured or formulated in the country and exported is up-to-dateimplement a procedure to check whether an export notification has been sent for the first export to a Party in the calendar yearfor chemicals in Annex III and those that are banned or severely restricted domestically, ensure that labelling provisions of the Convention are met and that the information is provided to accompany exports including Harmonized System Custom codes and safety data sheets.Countries have taken different approaches at national level to implement the provisions of the Convention for the PIC procedure and for the provisions concerning export notification and the information to accompany exported chemicals. One example has been to establish a permit system for the import or export of certain chemicals, e.g. those chemicals in Annex III, as well as those chemicals banned or severely restricted in the country. In such cases, the permits may be issued by the DNA and included in the shipping documents that are to be checked by Customs officers.
In doing so, DNAs must ensure that permits for the import or export of Annex III chemicals are only issued in compliance with the relevant import decisions.
Similarly, permits for the export of these chemicals are only issued upon confirmation that the provisions of the Convention, concerning export notification and the information to accompany exported chemicals such as labelling requirements or safety data sheet, have been met.
In such cases, there are three principal tasks for Customs officers:
identify the chemical in questionif the chemical falls within the scope of the Rotterdam Convention (PIC procedure or banned or severely restricted nationally) allow shipment only upon the issuance of a permit by the DNA; andin the absence of a permit from the DNA, Customs officers should stop the shipment and contact the DNA.7.3 Non-Governmental Stakeholders
The non-governmental stakeholders involved in the implementation of the Convention will vary among Parties. They might be considered to include associations of chemical manufacturers or formulators, commodity associations (including farmers associations or lobby groups), trade unions and environmental or consumer safety groups or other representatives of civil society with an interest or role in chemicals management.
The information available through the Convention such as improved labelling and the provision of safety data sheets with individual shipments of chemicals should help to reduce risks to human health and the environment from hazardous chemicals and should be made available to the relevant stakeholders.
In the implementation of the Convention it is also important to ensure that the national chemical industry, including manufacturers, formulators and traders, are aware of the provisions of the Convention and the possible impacts on their activities. In particular, with respect to the PIC procedure, DNAs should ensure they communicate to those stakeholders the national import decisions as well as those of other Parties. Chemical industry representatives should also be kept informed of developments in the Convention, in particular the listing of additional chemicals in Annex III. Such groups may also need to be consulted as part of the process of taking decisions on future import of chemicals.
The procedures for severely hazardous pesticide formulations set out in Article 6 clearly state that in developing a proposal for a severely hazardous pesticide formulation a Party may draw upon technical expertise from any relevant source. This could include groups or individuals involved in monitoring the health of workers that use pesticides, trade unions or others. Other sources of technical expertise may include environmental non-governmental organizations and farmers groups.
Click on the title to go back to the
Guidance
Home page
Index Menu